Introducing the gammaCore Sapphire™ device
gammaCore™ Sapphire, is a US FDA certified device, delivers non-invasive neuromodulation by stimulating the cervical branch of the vagus nerve. This rechargeable hand-held device includes:
- User selected intensity levels for personalized treatments.
- Up to 24 stimulations per day, so you can treat multiple attacks.
- 93 consecutive treatment days, enough treatments for 3 full months (treatment days may vary depending on model).
- Refill cards for your device are available to extend your treatment period.
- 36 month device option for chronic use.
Understanding the unpredictable nature of migraine and cluster headaches, the gammaCore™ Sapphire offers a flexible and sustainable solution with its 3 Month, 12 month, and 36 month refill kits. This range of options caters to the diverse needs of individuals, ensuring that everyone can find a treatment plan that aligns with their lifestyle and severity of their condition. Whether you're seeking to proactively manage your headache symptoms or need a reliable solution for sudden, acute attacks, gammaCore™ Sapphire stands ready to support your journey towards pain relief.
The availability of different refill kit durations empowers users to customize their treatment plan according to their specific requirements. For those who experience headaches sporadically, the 3 Month refill kit provides a convenient, short-term solution without committing to a longer duration. Meanwhile, the 12 Month kit is ideal for individuals who have identified a pattern in their headache occurrences and seek a year-long buffer against discomfort. Finally, the 36 Month option offers unmatched peace of mind for those battling chronic headache conditions, ensuring a long-term defense against pain.
Each refill kit ensures that your gammaCore™ Sapphire device remains a constant companion in your quest for relief, ready to preempt the onset of headache pain or intervene at the first sign of an attack. This flexibility in treatment duration not only demonstrates gammaCore™ Sapphire's commitment to accommodating the fluctuating nature of headache disorders but also underscores the device's role as a cornerstone in the management of such conditions.
By offering 3 Month, 12 month, and 36 month refill kits, gammaCore™ Sapphire underscores its dedication to providing sustained, adaptable support for individuals navigating the challenges of migraine and cluster headaches. With gammaCore™ Sapphire, you're not just investing in a device; you're embracing a lifestyle of preparedness and resilience, equipped to face headache pain with confidence and control.
NON-INVASIVE VAGUS NERVE STIMULATION

How gammaCore Sapphire™ works
By utilizing the innovative gammaCore™ Sapphire device, you can experience relief from migraine and cluster headache pain without the need for pills, injections, or medication-induced side effects. Unlike other treatments, such as implanted vagus nerve stimulators, gammaCore™ Sapphire is a non-invasive option that specifically activates the cervical branch of the vagus nerve. This cutting-edge technology stimulates the nerve through gentle electrical impulses, promoting healing and restoring balance to your body.
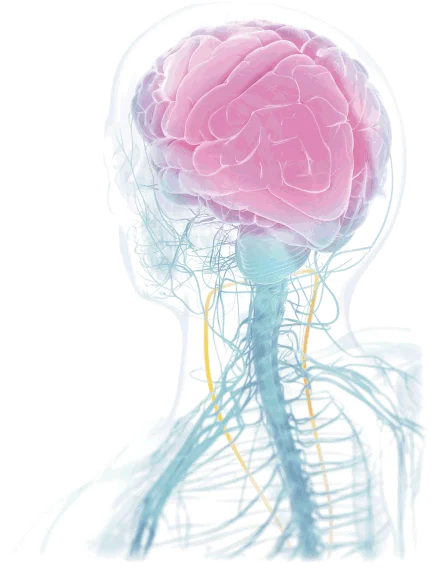
As the longest cranial nerve in the human body, the vagus nerve extends from the brain stem, traversing the neck as it connects the chest and abdomen. During gammaCore™ Sapphire treatments, this revolutionary device can be conveniently placed on either side of the neck, precisely targeting the location of the vagus nerve. By emitting gentle electrical impulses through the skin, gammaCore™ Sapphire stimulates the vagus nerve, promoting a sense of healing and restoring balance to the body.
Non-invasive and low risk
Simple and user-friendly, with numerous therapeutic advantages. No possibility of excessive usage and can be combined with other medications as required.
Portable
gammaCore™ Sapphire is a convenient, handheld device that brings relief wherever you are – whether you're at home or on the go. Its portable design allows you to easily operate it during your daily activities, ensuring that you can take control of your migraine or cluster headache pain whenever and wherever you need to.

Clinical practice
Demonstrated efficacy in multiple clinical trials. Well tolerated with less medication-like side effects; can be used up to 24 times per day.
DISCOVER THE SIMPLICITY OF USING THE GAMMACORE™ SAPPHIRE
The device is incredibly user-friendly, making it effortlessly easy to operate. Its intuitive design and straightforward functionality ensure that anyone can use it with ease. Whether you are a tech-savvy individual or someone who prefers simplicity, the gammaCore™ Sapphire device caters to all users. With just a few simple steps, you can experience the remarkable relief it provides. Say goodbye to complex instructions or confusing settings – this device simplifies the process, allowing you to focus on finding relief from your migraine or cluster headache pain.
Explore the Quick Start Guide for an effortless and seamless experience with the gammaCore™ Sapphire.
Intended use
gammaCore™ Sapphire is indicated for the treatment and/or prevention of primary headache (migraine, cluster headache and hemicrania continua) and medication overuse headache in adults.
SUPPORTED BY SCIENTIFIC EVIDENCE
Vagus nerve stimulation has undergone extensive research and study, resulting in the gammaCore™ Sapphire device receiving 7 different FDA clearances. This impressive device is supported by robust scientific and clinical data, including 7 randomized controlled trials and over 40 peer-reviewed clinical papers. With more than 30 mechanisms of action peer-reviewed papers available, you can dive into the wealth of information surrounding the safe and effective use of the gammaCore™ Sapphire device. Read these papers for more information.
- Non-invasive neuromodulation of the cervical vagus nerve in rare primary headaches
- Evidence of activation of vagal afferents by non-invasive vagus nerve stimulation: An electrophysiological study in healthy volunteers
- Noninvasive vagus nerve stimulation as acute therapy for migraine: The randomized PRESTO study
- Vagus nerve stimulation for prevention of migraine: The multicenter, randomized, double-blind, sham-controlled PREMIUM II trial
- NICE Guideline on transcutaneous stimulation of the cervical branch of the vagus nerve for cluster headache and migraine
Ready to get started or learn more?
Just fill in the fields below and click submit
Important Safety Information
gammaCore Sapphire™ (non-invasive vagus nerve stimulator) is intended to provide non-invasive vagus nerve stimulation (nVNS) on the side of the neck. gammaCore is indicated for:
- The preventive treatment of migraine headache in adolescent (age 12 and older) and adult patients.
- The acute treatment of pain associated with migraine headache in adolescent (age 12 and older) and adult patients.
- Adjunctive use for the preventive treatment of cluster headache in adult patients.
- The acute treatment of pain associated with episodic cluster headache in adult patients.
- Treatment of hemicrania continua and paroxysmal hemicrania in adults.
- The effectiveness of gammaCore has not been established in the acute treatment of chronic cluster headache
- gammaCore is contraindicated for patients if they:
- Have an active implantable medical device, such as a pacemaker, hearing aid implant, or any implanted electronic device
- Are using another device at the same time (e.g., TENS Unit, muscle stimulator) or any portable electronic device
- Safety and efficacy of gammaCore have not been evaluated in the following patients:
- Patients diagnosed with narrowing of the arteries (carotid atherosclerosis)
- Patients with a metallic device, such as a stent, bone plate, or bone screw, implanted at or near the neck
- Patients who have had surgery to cut the vagus nerve in the neck (cervical vagotomy)
- Pediatric patients (younger than 12 years)
- Pregnant women
- Patients with clinically significant hypertension, hypotension, bradycardia, or tachycardia
NOTE: This list is not all inclusive. Please refer to the gammaCore Instructions for Use for all of the important warnings and precautions before using or prescribing this product.











